The Universiti Malaya Healthcare Data Safe Haven (UM-HDSH) is a strategic initiative to enable the secure and ethical use of sensitive clinical data generated at the University Malaya Medical Centre (PPUM/UMMC). As healthcare increasingly depends on data to inform diagnostics, clinical decision support, public health planning, and AI-enabled applications, the project addresses a critical need to harness real-world clinical data without compromising patient privacy or trust.
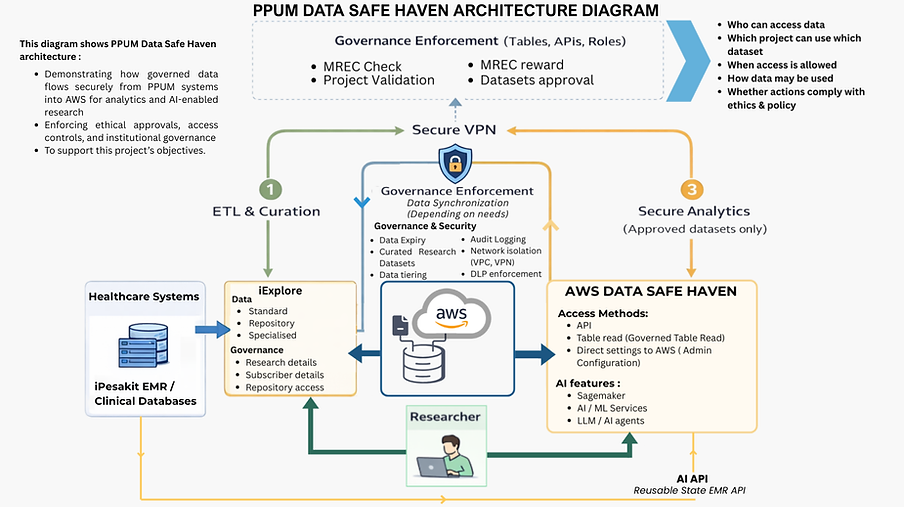
Anchored within PPUM, the Data Safe Haven operates as a controlled repository and analysis environment for patient-sensitive data collected through hospital electronic medical record systems such as iPesakit and iExplore. Clinical data are curated and rigorously anonymised before being made accessible to approved researchers under strictly regulated conditions. All analyses are conducted within the secure environment, with no downloading or external transfer of data permitted. This enables meaningful clinical and public health research while safeguarding confidentiality.
A defining feature of the UM-HDSH is its robust governance framework, which integrates institutional oversight, ethics approval, clinician involvement, and clear policies on data ownership and intellectual property. Data access is project-specific, time-limited, and fully auditable, ensuring transparency, accountability, and compliance with Malaysian data protection regulations. This governance-first approach supports long-term, sustainable stewardship of clinical data.
The project was developed in collaboration with Amazon Web Services (AWS), which supported the configuration of a secure, cloud-based proof-of-concept system. As part of its initial implementation, the Data Safe Haven will be piloted using the PPUM Breast Cancer Registry, serving as a case study to demonstrate patient survival prediction and to seed future AI-enabled clinical workflows.
Deployed initially at PPUM, the UM-HDSH functions as a testbed for responsible clinical data use, with clear potential for expansion across additional disease registries and, subject to regulatory alignment, broader national adoption.